Details of a chemical method for rejuvenating an old batteryYou have probably never stopped to consider it, but very few batteries are replaced because they stop working. In almost every case, the battery is replaced because it doesn't work well enough any more. What is not generally realised is that something can be done at this stage which may well prolong the useful life of the battery - quite often by many years, and that the same treatment carried out earlier may well have stopped the symptoms from occurring in the first place.
The reason for a battery failing to work properly anymore is due to the chemical processes which take place within each cell. As the battery discharges, the +ve. plate reacts with the sulphuric acid to produce lead ions and water. The -ve. plate dissolves to form lead ions in the process, which finally leads to the battery's complete failure. On charging, the +ve. plate builds up a thicker coating of lead oxide, removing water and lead ions from the sulphuric acid as it does so. The -ve. plate fizzes and releases hydrogen from the sulphuric acid as it builds up a coating of lead. It is the lead ions formed in the discharge cycle which cause problems. They combine with sulphate ions in sulphuric acid to form highly insoluble lead sulphate. When this coats the plates of the battery, it fails to deliver enough power to be of use although it may well be thoroughly serviceable in every other way; only the 'sulphating' stops the battery delivering enough power to start the vehicle.
As can be seen from the above, treating a battery with EDTA is likely to be most effective when the battery, for one reason or another, spends periods when it is not fully charged, and so contains too many lead ions. This is likely to occur if the vehicle is used just for short trips, is infrequently used, or suffers from an inefficient dynamo or alternator. To treat a battery with EDTA you add about a rounded teaspoonful of the powder to each cell - this assumes an average sized battery, but the exact amount is in no way critical. You should use the vehicle normally for a while or agitate the battery frequently for a few days, then give it a thorough charge to build up on the cleaned plate areas. On the assumption that sulphating has been affecting the performance of your battery, an increased performance will be noted from here on. Thanks to the MMOC. EDTA is available online from Pure Research - http://www.pureresearch.co.uk/ |
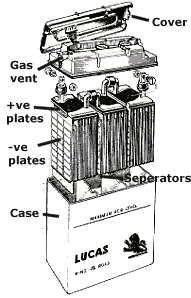
 To understand what can be done requires a simple appreciation of how a lead acid
battery works. These batteries are a series of cells; the most common is the
1
2V type
which is six cells in series. Each cell consists of a plate of lead and a
supported plate of
lead (IV) oxide, both immersed in fairly concentrated sulphuric acid. The lead
oxide
plate is the +ve. pole, the lead plate is the -ve. pole. As the battery
discharges, electrons
flow from the -ve. to the +ve. pole; when we charge the battery, we just drive
electrons
back in the opposite direction.
To understand what can be done requires a simple appreciation of how a lead acid
battery works. These batteries are a series of cells; the most common is the
1
2V type
which is six cells in series. Each cell consists of a plate of lead and a
supported plate of
lead (IV) oxide, both immersed in fairly concentrated sulphuric acid. The lead
oxide
plate is the +ve. pole, the lead plate is the -ve. pole. As the battery
discharges, electrons
flow from the -ve. to the +ve. pole; when we charge the battery, we just drive
electrons
back in the opposite direction.
 The sulphating can effectively be removed or prevented by adding to each cell a
weak
organic acid, ethylene diamine tetraacetic acid, known to chemists as EDTA. It
is a
complex analytical reagent which forms co-ordination compounds with many metal
ions,
including the lead ions formed in the discharge cycle of a battery. The compound
formed by lead ions and EDTA is very stable in alkaline solution but not in the
acidic
medium of a battery: this is extremely fortunate. As EDTA forms a compound with
the
lead ions in the lead sulphate on the battery plate, it breaks down again. EDTA
and lead
sulphate are regenerated, but this time the lead sulphate doesn't coat the
plate, it sinks
down to the bottom of the cell where it lies harmlessly since it doesn't conduct
electricity, and the EDTA is free to continue its work. What EDTA effectively
does then,
is free the plates of sulphating.
The sulphating can effectively be removed or prevented by adding to each cell a
weak
organic acid, ethylene diamine tetraacetic acid, known to chemists as EDTA. It
is a
complex analytical reagent which forms co-ordination compounds with many metal
ions,
including the lead ions formed in the discharge cycle of a battery. The compound
formed by lead ions and EDTA is very stable in alkaline solution but not in the
acidic
medium of a battery: this is extremely fortunate. As EDTA forms a compound with
the
lead ions in the lead sulphate on the battery plate, it breaks down again. EDTA
and lead
sulphate are regenerated, but this time the lead sulphate doesn't coat the
plate, it sinks
down to the bottom of the cell where it lies harmlessly since it doesn't conduct
electricity, and the EDTA is free to continue its work. What EDTA effectively
does then,
is free the plates of sulphating.